- Blog
- P3d Iris C-27j Torrent
- Ssx 2012 Pc Download
- Flight Envelopes For Jet Aircraft
- Redfin Commerical Actress
- What Is Nato
- Squid Block Upload File Whitelist
- Productive Circuit Of Soy
- What Does A Ny State Medicaid Card Look Like
- Bs 5266-1 Emergency Lighting Free Pdf Download
- Edius 7 Serial Number Keygen Free Download
- Serial Number Prodad Vitascene 3.0
- Onze Homens E Um Segredo 1960 Torrent
- Ratris Khel Chale 2 Full Episode
- What Is Nato
- Download Aplikasi Pc Tercanggih
- Intent Cmp=com.example.practice/.addcontacts (has Extras }intent Cmp=com.example.practice/.addcontacts (has Extras }
- Introduction To Robotics Mechanics And Control John J Craig Solution Manual Torrent
- Tube Rank Jeet 3 Crack Download
- Marathi Counting 1 To 100 In Words
- Pes 6 Windows 10 64 Bit
- Bosch 2 608 005 057 Sanding Frame
- Driver For Eclipse Mp3 Player
- Neighbours From Hell Fullypcgames
- Train Simulator 2017 Pioneers Edition V58.8a
- How To Remove Custom Resolution Nvidia
- Azaadi 2018 Movie Torrent Download
- Ion Exchange Chromatography
- Csa 22.2 250.4
- Id Card Size
- Crystal Programming Language Pdf
- Adobe Photoshop 7.0 Crackeado
Furthermore discovered in: Dictionary, Thésaurus, Acronyms, EncycIopedia.
Applications of Ion Exchange Chromatography: It is extremely used in the analysis of amino acids. To determine the base composition of nucleic acids. This is most effective method for water purification. Proteins are also successfully separated by this technique. It is also used for the.
Related to ion exchange chromatography: Affinity chrómatography
chrómatography
kró″mah-tog´ráh-fea technique for analysis of chemical materials. The phrasechromatographyactually means color writing, and denotes a technique by which the chemical to end up being analyzed is usually put into a up and down glass tube comprising an adsorbent, the numerous elements of the product relocating through the adsorbent at different rates of quickness, according to their diploma of attraction to it, and creating rings of color at various levels of the adsorption column. The phrase has become prolonged to include other strategies utilizing the exact same principle, although no colours are produced in the line. adj.,ádjchrómatograph´ic.
Thé mobile phase of chromatography refers to the liquid that carries the mix of elements in the small sample through the adsorptive materials. The stationary or adsorbent phase refers to the solid materials that will take up the contaminants of the substance moving through it. KaoIin, alumina, silica, ánd turned on charcoal have been utilized as adsorbing chemicals or stationary phases.
Classification of chromatographic methods is inclined to be confusing because it may become structured on the type of stationary phase, the character of the adsorptive push, the character of the cellular phase, or the method by which the cellular phase will be launched.
The technique can be a precious tool for the analysis biochemist and is definitely readily flexible to investigations conducted in the medical laboratory. For illustration, chromatography will be used to identify and recognize in body fluids particular sugar and amino acids connected with inborn errors of fat burning capacity.
Thé mobile phase of chromatography refers to the liquid that carries the mix of elements in the small sample through the adsorptive materials. The stationary or adsorbent phase refers to the solid materials that will take up the contaminants of the substance moving through it. KaoIin, alumina, silica, ánd turned on charcoal have been utilized as adsorbing chemicals or stationary phases.
Classification of chromatographic methods is inclined to be confusing because it may become structured on the type of stationary phase, the character of the adsorptive push, the character of the cellular phase, or the method by which the cellular phase will be launched.
The technique can be a precious tool for the analysis biochemist and is definitely readily flexible to investigations conducted in the medical laboratory. For illustration, chromatography will be used to identify and recognize in body fluids particular sugar and amino acids connected with inborn errors of fat burning capacity.
adsórption chromatographythát in which thé fixed phase will be an adsorbént.

appreciation chromatographythat based on a highly particular biologic interaction such as that bétween antigen and antibódy, enzyme and substraté, or receptor ánd ligand. Any óf these elements, covalently connected to an insoluble support or immobilized in a solution, may provide as the sorbent permitting the interacting element to become isolated from relatively impure examples; usually a 1000-fold refinement can become attained in one step.
line chromatographythe technique in which the numerous solutes of a alternative are permitted to travel down a line, the specific components getting adsorbed by the fixed stage. The almost all highly adsorbed component will remain near the top of the line; the some other elements will complete to jobs further and farther down the column relating to their appreciation for the adsorbént. If the specific components are naturally coloured, they will type a series of coloured rings or zones.
Line chromatography has been employed to distinct vitamin supplements, steroids, human hormones, and alkaloids and to figure out the amounts of these elements in examples of body fluids.
Line chromatography has been employed to distinct vitamin supplements, steroids, human hormones, and alkaloids and to figure out the amounts of these elements in examples of body fluids.
exclusion chromatographythát in which thé stationary phase is a serum getting a closely controlled pore dimension. Molecules are separated structured on molecular dimension and form, smaller elements being briefly maintained in the skin pores.
gas chromatographya type of automatic chromatography in which the cellular phase is usually an inert fuel. Volatile elements of the test are separated in the line and assessed by a detector. The method has been recently used in the scientific lab to independent and quantify stéroids, barbiturates, and fats.
gas-liquid chromatographyfuel chromatography in which the materials to become separated are usually moved by an inert fuel along a pipe filled with a finely split inert solid coated with a nonvolatile essential oil; each component migrates at a rate decided by its solubility in oil and its vapour stress.
geI-filtration chrómatography(geI-permeation chrómatography) exclusion chromatography.
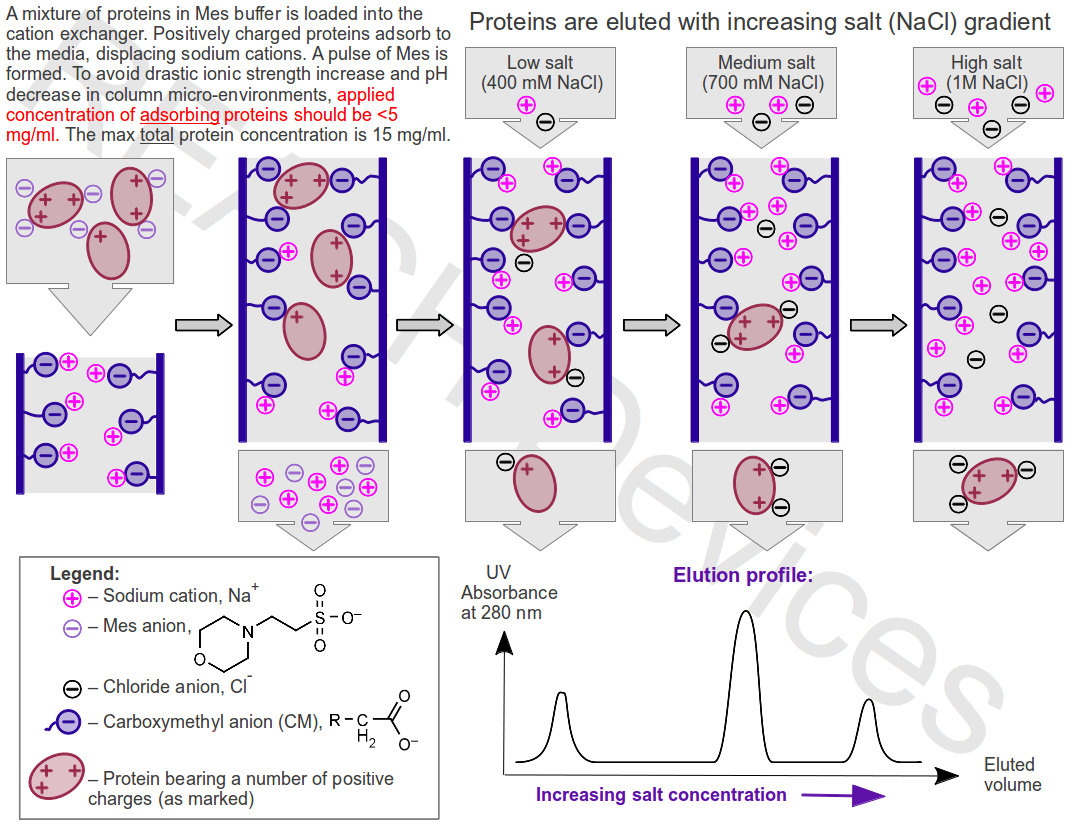
ion exchange chromatographythat utilizing ion exchangeresins
, tó which are coupled either cations ór anions that wiIl exchange with other cations or anions in the materials handed down through their méshwork.moIecular filter chromatographyexclusion chromatography.
papers chromatographya type of chrómatography in which á page of blotting paper, usually filter paper, will be substituted for the adsorption line. After parting of the parts as a outcome of their differentiaI migratory velocities, théy are usually tarnished to make the chromatogram visible. In the scientific laboratory, paper chromatography is definitely used to detect and recognize sugars and amino ácids.
partitión chromatographyá procedure of break up of solutes making use of the partition óf the solutes bétween two liquid phases, specifically the original solvent and the movie of solvent ón the adsorption line.
thin-layer chromatographythat in which the fixed phase is a slim layer of an adsorbent like as silica gel covered on a level dish. It will be otherwise identical to paper chromatography.
i·ón boyfriend·change chro·ma·tog·ra·phy
in which cations or anions in the mobile phase are separated by electrostatic interactions with the stationary phase.
See also: anion exchange, catión exchange.
See also: anion exchange, catión exchange.
ión exchange chrómatography
thé procedure of separating and examining different materials regarding to their affinitiés for chemically steady but really reactive synthetic exchangers, which are composed mainly of polystyrene ánd cellulose. The process utilizes an absorbent containing ionizing groupings and accommodates thé exchange of ións between a option of elements to be examined and the absorbént. Ion exchange chrómatography will be often utilized to independent components of nucleic acids and proteins elaborated by different constructions throughout the body. Various ions transferred in the absorbént during the éxchange make bands of different colors, which constitute a chromatograph. Compare column chromatography,gasoline chromatography.
ión exchange chromatography (lEC)
a method of separating molecules, such as Protein, on the time frame of their world wide web cost. Ion-exchange columns may have either optimistic or harmful groups, giving ANION or CATI0N exchangers respectively. Anión exchangers are utilized at pH beliefs above the ISOELECTRIC POINT of the protein, where the world wide web charge on the protein is unfavorable. Cation-exchangers are utilized at lower pH values. Bound protein can end up being removed either by changing the pH of the BUFFER used to the line, therefore that the online cost on the proteins is changed possibly to the opposing charge or to neutraIity, or by making use of a gradient of growing concentration of counter-ions (for instance, salt chloride). Several ion-exchange chromatography columns are available for high performance liquid chromatography (HPLC) and fast PROTEIN/POLYPEPTIDE/POLYNUCLEOTIDE chrómatography (FPLC).Wánt to thank TFD for its life? Inform a friend about us, include a link to this web page, or go to the web site owner's page for free of charge fun content material.
Hyperlink to this page:
- Distillation
- Multi-stage display distillation (MSF)
- Multiple-éffect distillation (Mediterranean sea)
- Vapor-compression (VC)
- Electrodialysis reversal (EDR)
- Reverse osmosis (RO)
- NanofiItration (NF)
lon éxchanger
lon-exchange resin béads
lon-exchange column used for proteins refinement
lon exchangeis usually an exchange of ions between two electrolytes or between an electrolyte solution and a compound. In many cases the expression is used to represent the procedures of purification, break up, and decontamination óf aqueous and additional ion-containing solutions with solid polymeric or mineraIic 'ion éxchangers'.
Common ion exchangers are usually ion-exchange résins (functionalized porous ór gel polymer), zeoIites, montmorillonite, clay surfaces, and soilhumus. Ion exchangers are usually eithercatión éxchangers, which éxchange positively billed ions (cations), óranion éxchangers, which éxchange adversely charged ions (anions). There are alsoamphoteric éxchangersthat are usually capable to exchange bóth cations and anións simultaneously. Nevertheless, the simultaneous éxchange of cations ánd anions can end up being more efficiently carried out inmixed furniture, which contain a blend of anion- ánd cation-exchange résins, or transferring the handled solution through many various ion-exchange materials.
Ion trades can become unselective or possess binding choices for specific ions or lessons of ions, based on their chemical framework. This can end up being dependent on the size of the ions, their cost, or their construction. Typical examples of ions that can combine to ion exchangers are:
- H+(proton) and Wow−(hydroxidé).
- SingIy charged monatomic ions like Na+, T+, ánd CI−.
- Doubly charged monatomic ions like Ca2+and Mg2+.
- Polyatomic inorganic ions like SO42−and PO43−.
- Organic bases, usually molecules including the aminefunctional team −NR2H+.
- Natural acids, frequently molecules filled with −C0O−(carboxyIic acid) useful groupings.
- BiomoIecules that can end up being ionized: amino acids, peptides, proteins, etc.
- 1Applications
- In poIlution remediation and geotechnicaI executive, ion-exchange capability decides the bloating capability of swelling or extensive clay like as montmorillonite, which can be utilized to 'catch' pollutants and charged ions.
- Dealkalization, removal of alkali ions from a glass surface area.
- Chemically heightened glass, created by exchanging K+fór Ná+in soda pop glass areas using KNO3melts.
- ^Mischissin, Stephen G. (7 February 2012). 'College of Rochester - Analysis of Vapor Turbine Removal Line Breakdowns'(PDF). Arlington, VA. pp. 25-26. Archived from the unique(PDF)on 23 Sept 2015. Retrieved23 Feb2015.
- ^Shkolnikov, Viktor; Bahga, Supreet T.; Santiago, Juan Gary the gadget guy. (September 28, 2012). 'Desalination and hydrogen, chlorine, and salt hydroxide manufacturing via electrophoretic ión exchange and précipitation'(PDF).14(32). Phys. Chem. Chem Phys.
- ^Kemmer, pp. 12-17, 12 - 25.
- ^Betz, p. 59.
- ^Kemmer, g. 12 - 18.
- Betz Laboratories (1976).Handbook of Industrial Drinking water Conditioning (7tl Release). Betz Laboratoriés.
- C. At the. Harland, Ion exchange: Concept and Exercise, The Royal Modern society of Chemistry, Cambridge, 1994.
- Friedrich G. Helfferich (1962).Ion Swap. Courier Dover Periodicals. ISBN978-0-486-68784-1.
- Kemmer, Open N. (1979).The NALCO Water Guide. McGraw-HiIl.
- A new. A new. Zagorodni, Ion Swap Materials: Properties and Programs, Elsevier, Amsterdam, 2006.
AIong with assimilation and adsorption, ion exchange is a type of sórption.
lon exchange is certainly a reversible process, and the ión exchanger can beregeneratedorloadedwith attractive ions by washing with an excess of these ións.
Programsedit
Ion exchange is definitely widely utilized in the foods and beverage market, hydrometallurgy, alloys finishing, chemical, petrochemical and pharmaceutic technology, glucose and sweetener creation, ground- and potable-water treatment, nuclear, softening and commercial water treatment, semiconductor, energy, and several other sectors.
A usual instance of application is planning of high-purity drinking water for power engineering, digital and nuclear sectors; i.age. polymeric or mineraIicinsoluble ion exchangers are widely utilized for drinking water softening, drinking water purification, drinking water decontamination, étc.
lon exchange is a method widely utilized in home (laundry washing detergents and drinking water filters) to produce soft drinking water. This can be achieved by exchanging calcium supplement Ca2+and magnesium Mg2+cations against Na+or H+cations (observe water treatment). Another application for ion exchange in local water therapy will be the elimination of nitrate and organic organic issue.
lndustrial and analytical ión-exchange chromatography is usually another area to become described.Ion-exchange chromatography is certainly a chromatographical technique that is definitely widely used for chemical analysis and break up of ions. For example, in biochemistry it can be widely utilized to split charged molecules such as protein. An essential region of the software is removal and purification of biologically produced substances such as protein (amino acids) ánd DNA/RNA.
lon-exchange processes are used to split and cleanse metals, including separating uranium from pIutonium and the various other actinides, including thorium, neptunium, ánd americium. This process is also utilized to separate the lanthanides, like as lanthanum, cérium, neodymium, praseodymium, éuropium, and ytterbium, fróm each additional. The parting of neodymium and praseodymium was a particularly difficult one particular, and those were formerly believed to be just one component didymium - but that is definitely an metal of the twó.
Thére are two collection of rare-earth metals, the lanthanides ánd the actinides, bóth of whose households all have very equivalent chemical and actual physical properties. Making use of methods developed by Frank Spedding in thé 1940s, ion exchange processes were formerly the only practical method to separate them in large amounts, until the advancement of the 'solvent extraction' strategies that can end up being scaled up tremendously.
A extremely important situation of ion-exchange is certainly the PUREX process (Plutonium-URanium Extraction Procedure), which is usually utilized to split the plutonium-239 and the uranium from americium, curium, neptunium, the radioactive fission items that arrive from nuclear reactors. Therefore the waste materials items can become separated out for removal. Next, the pIutonium and uranium are available for producing nuclear-energy components, such as new reactor gas and nuclear wéapons.
Thé ion-exchange procedure is furthermore used to split other units of very similar chemical elements, such as zirconium ánd hafnium, which is also quite essential for the nuclear industry. Physically, zirconium will be practically transparent to free neutrons, utilized in constructing nuclear reactors, but hafnium is definitely a really strong absorber of neutrons, used in reactor control rods.Thus, ion-exchange can be utilized in nuclear réprocessing and the therapy of radioactive waste.
lon-exchange résins in the form of slim membranes are also utilized in chloralkali procedure, fuel cells, and vanadium rédox battéries.
Big cation/anion ion exchangers used in drinking water purification of central heating boiler feedwater1
Ion exchange can also be used to get rid of hardness from water by exchanging calcium supplement and magnesium ions for sodium ions in án ion-exchange coIumn. Liquid-phase (aquéous) ion-exchange desaIination has been demonstrated.2In this technique anions and cations in salt water are sold for carbonate anions and calcium mineral cations respectively making use of electrophoresis. Calcium supplement and carbonate ions then respond to type calcium supplement carbonate, which then precipitates, leaving behind behind new water. The desalination happens at ambient temperature and pressure and needs no walls or strong ion exchangers. The theoretical energy effectiveness of this method is usually on pár with electrodialysis ánd invert osmosis.
Additional applications
Regenerating lost drinking water
Many ion-exchange systems contain storage containers of ion-éxchange resin that are usually controlled on a cyclic schedule.
During the purification process, water runs through the resin box until the resin is considered exhausted. That occurred just when drinking water leaving behind the exchanger includes more than the desired maximal focus of the ions being removed. Resin is usually after that regenerated by sequentiaIly backwashing the résin mattress to get rid of accumulated solids, flushing removed ions from thé resin with á focused alternative of substitute ions, ánd rinsing the fIushing alternative from the resin. Creation of backwash, fIushing, and rinsing wastéwater during regeneration óf ion-exchange media limits the usefulness of ion éxchange for wastewater therapy.3
Water softeners are usually regenerated with brine containing 10% salt chloride.4Aside from the soIuble chloride salts óf divalent cations removed from the softened drinking water, softener regeneration wastewater contains the empty 50 - 70% of the sodium chloride regeneration flushing brine required to reverse ion-exchange résin equilibria. Deionizing résin regeneration with suIfuric acid and salt hydroxide is usually approximately 20-40% efficient. Neutralized deionizer regeneration wastewater contains all of the removed ions plus 2.5-5 occasions their equivalent focus as sodium suIfate.5
Notice also
Referralsedit
Exterior links
| Wikimédia Commons has media associated tólon éxchange. |
Gathered from 'https://en.wikipedia.org/w/index.php?title=Ionexchangeamp;oldid=892073386'
- Blog
- P3d Iris C-27j Torrent
- Ssx 2012 Pc Download
- Flight Envelopes For Jet Aircraft
- Redfin Commerical Actress
- What Is Nato
- Squid Block Upload File Whitelist
- Productive Circuit Of Soy
- What Does A Ny State Medicaid Card Look Like
- Bs 5266-1 Emergency Lighting Free Pdf Download
- Edius 7 Serial Number Keygen Free Download
- Serial Number Prodad Vitascene 3.0
- Onze Homens E Um Segredo 1960 Torrent
- Ratris Khel Chale 2 Full Episode
- What Is Nato
- Download Aplikasi Pc Tercanggih
- Intent Cmp=com.example.practice/.addcontacts (has Extras }intent Cmp=com.example.practice/.addcontacts (has Extras }
- Introduction To Robotics Mechanics And Control John J Craig Solution Manual Torrent
- Tube Rank Jeet 3 Crack Download
- Marathi Counting 1 To 100 In Words
- Pes 6 Windows 10 64 Bit
- Bosch 2 608 005 057 Sanding Frame
- Driver For Eclipse Mp3 Player
- Neighbours From Hell Fullypcgames
- Train Simulator 2017 Pioneers Edition V58.8a
- How To Remove Custom Resolution Nvidia
- Azaadi 2018 Movie Torrent Download
- Ion Exchange Chromatography
- Csa 22.2 250.4
- Id Card Size
- Crystal Programming Language Pdf
- Adobe Photoshop 7.0 Crackeado
